
Oncology

This week, HistoSonics announced it will bring its ultrasound system for destroying liver tumors into VA hospitals, Edwards Lifesciences reports encouraging TRISCEND II trial results at TCT, study finds blood test for CRC screening are less cost-effective than alternatives, and more.
Medtech Insight talked with GE HealthCare’s chief AI officer Parminder “Parry” Bhatia at HLTH about the firm’s new CareIntellect for Oncology offering to help clinicians make efficient use of multimodal patient data, his vision for projects within AI Innovation Lab, and the future of AI in health care.

University of Cambridge's soon-to-be spun-out Nimble Genomics is among grant-winning projects in Capital Enterprise's Cancer Tech Accelerator. Medtech Insight spoke to clinical oncologist and Nimble founder Henno Martin and co-founder Radek Lach to learn more what about the brain cancer blood test’s prospects in an increasingly competitive liquid biopsy space.
At HLTH 2024, GE HealthCare unveiled CareIntellect for Oncology, a generative AI-driven platform to help oncologists quickly assess patient data across multiple sources to save time and improve treatment planning. The platform, currently in evaluation at two US hospitals, is set for launch in the second half of 2025.

Medtech Insight was on the ground at HLTH looking for innovative companies. Learn about seven start-ups using AI to help physicians detect conditions including prostate and breast cancers, seizures and heart failure; assess patients for cognitive decline validate and deploy algorithms, and monitor patients in and out of the hospital.

This week, Hologic announced it would pay $350m for uterine fibroid treatment firm Gynesonics; the US FDA authorized a Novocure cancer treatment and a surgical robot from CMR Surgical; and the IMDRF announced 15 new members.

This week, J&J announced that it was buying heart failure device firm V-Wave; Procept got the FDA’s OK on a clinical trial of its Aquablation treatment for prostate cancer; and CMS began to consider Medicare reimbursement of Abbott’s TriClip tricuspid repair device.

The College of American Pathologists wants a US district court to scrap the FDA’s final rule that unilaterally assumes oversight of lab-developed tests, but not because the college feels the agency has no business regulating them. Helena Duncan, senior director of quality at CAP, explained the college’s position to Medtech Insight.

The Octane Medical Innovation Forum brought together industry experts, entrepreneurs and investors to discuss a range of topics. Medtech Insight was on the ground to bring some memorable perspectives from industry leaders.

The US FDA’s Office of Women’s Health provides a research roadmap to address health concerns specific to women. The FDA recently updated the roadmap, outlining areas in which further research is needed.

This week, Establishment Labs Holdings announced the FDA gave it premarket approval for Motiva breast implant, Cologuard lands FDA approval for Cologuard Plus and GE HealthCare gets FDA nod for a new imaging agent. The FDA announces another expansion for TAP into ophthalmology and radiology. The AAMI and CTA will join forces to develop standards for AI and ML-enabled health care products.
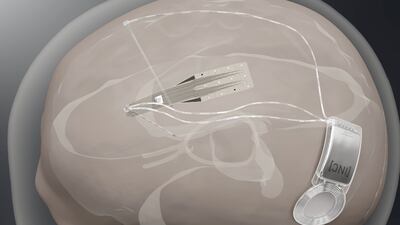
Spain-based Inbrain Neuroelectronics plans first-in-human study to show safety of its graphene-based technology in direct contact with human brain while also developing a second interface for treating Parkinson’s disease.

Biopharmaceutical giant AstraZeneca has partnered with start-up “unicorn” Owkin to develop an AI-powered tool to prescreen for gBRCA mutations on the basis of morphological features in digitized pathology slides. Built on extensive, high-quality data sourced from the France-based PortrAIt consortium, the AI will help to prioritize patients for further testing, streamlining the diagnostic process, Owkin says.

This week, Neuralink announced it received US FDA breakthrough device designation for a device to restore sight; medtechs Discure and DeepLook secured new funding; FDA pump recalls from B. Braun Medical and Fresenius Kabi; Axonics prevails in patent infringement lawsuit with Medtronic; Merit Medical buys Cook Medical for $210m.

Leaders from Eli Lilly and Vertex Pharmaceuticals, 9amHealth, Features Capital, and accelerator PharmStars discussed the convergence of biopharma, medtech and healthtech, and what is needed to achieve health care transformation, in an LSX World Congress USA panel.
Exact Sciences presented new data for its blood-based colorectal cancer screening test, which BTIG analysts say bodes well for the test’s probability to receive US FDA approval. Study results are expected in the first half of 2025.

The US FDA now requires mammograms include information about breast density, a significant factor that raises breast cancer risk and can hide tumors.

Highlights from Medtech Insight's on-the-ground coverage of LSX in Boston.

In a letter to US lawmakers, the Association for Diagnostics and Laboratory Medicine says the FDA’s final rule regulating lab-developed tests will stifle innovation and that Congress needs to step in and stop it.

With the reversal of the Chevron doctrine in June, lower courts will now have more say in deciding regulatory statutes when the language is murky. But will that open the door to more legal challenges from the healthcare industry against government regulations it finds unfavorable? A pair of legal experts recently discussed the potential implications of the Court’s decision.