ADVERTISEMENT
Approvals

Pipeline Watch is a weekly snapshot of selected late-stage clinical trial events and approvals announced by pharmaceutical and biotech companies at medical and industry conferences, in financial and company presentations, and in company releases and statements.

Alvotech and US marketing partner Teva have celebrated a further approval for their Selarsdi ustekinumab biosimilar rival to Stelara that encompasses a new presentation as well as a label expansion to include adults with Crohn’s disease and ulcerative colitis.

Pipeline Watch is a weekly snapshot of selected late-stage clinical trial events and approvals announced by pharmaceutical and biotech companies at medical and industry conferences, in financial and company presentations, and in company releases and statements.

Accord has received a nod from the EMA for its Stelara biosimilar, while Neuraxpharm has seen a buprenorphine hybrid film endorsed and Viatris has received a positive CHMP opinion for an eltrombopag generic.

Partners Dong-A ST and Meiji Seika Pharma have demonstrated their credentials in the US biosimilar market, bagging a US Food and Drug Administration approval for their biosimilar Stelara product that will be commercialized by Accord Healthcare.

The US FDA approved the drug for hemophilia A and B, and while it may struggle to compete in hemophilia A against Roche’s Hemlibra, it has a big convenience advantage in hemophilia B.

Pipeline Watch is a weekly snapshot of selected late-stage clinical trial events and approvals announced by pharmaceutical and biotech companies at medical and industry conferences, in financial and company presentations, and in company releases and statements.
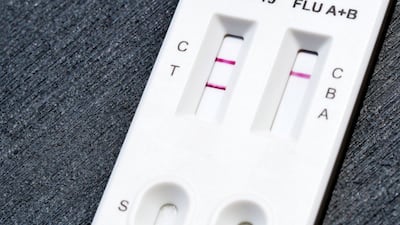
FDA grants de novo authorization to Healgen Rapid Check COVID-19/Flu A&B Antigen Test, making it the first OTC flu test to be cleared outside the emergency use pathway.

Richard Lowenthal, co-founder and CEO of ARS Pharmaceuticals, highlights the crucial unmet need for needle-free devices to treat type 1 allergic reactions, given challenges associated with current epinephrine injectors. Hear what’s next for the Neffy intranasal spray and ARS Pharmaceuticals.
While vatiquinone may have missed its primary endpoint in a Phase III trial, PTC is ready to file the drug for the debilitating, life-shortening disorder after showing that it slowed disease progression over 144 weeks.