Litigation
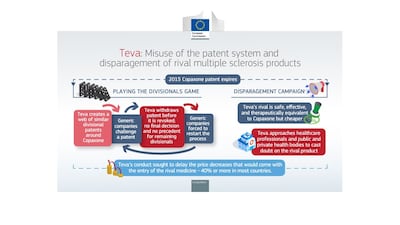
Teva has been fined €463m – just over half a billion US dollars – over a breach of EU antitrust rules, after the European Commission found that it abused its dominant position to delay competition to Copaxone, including by misusing the patent system and disparaging rivals. The firm has strongly disagreed with the decision – which is claims is “legally untested” and “not supported by the facts” – and says it will appeal.

Judges on the Third Circuit panel in the BMS and JNJ IRA cases seemed sympathetic to industry’s concerns about the government using the term “maximum fair price” in the IRA’s Medicare drug price negotiation program.

Multiple and repeated complaints will sharpen the Office of Prescription Drug Promotion’s focus on an advertisement, Director Catherine Gray said, while Foley Hoag partner August Horvath said the self-regulatory NAD process is best suited to complaints that lack a ‘great scientific basis’ for objecting.

The outcome of the November presidential election may impact whether the government is willing to defend FDA's relaxation of the mifepristone REMS. The case poses risks for the broader drug approval process.

With a single drug category and an uncertain political future, the FTC’s legal efforts may gain limited traction. Was focusing on insulin rebating a wisely targeted approach or did the case underemphasize the ‘emotionally more powerful consumer deception issue’?

Pink Sheet reporter and editors discuss an emerging pharma strategy to avoid Medicare price negotiations, legal wrangling related to compounding GLP-1 drugs for obesity and diabetes, and the varying opinions of FDA officials on the acceptability of artificial intelligence models that are not fully explainable.

FDA law experts do not buy the outsourcers’ argument that the agency must go through notice-and-comment rulemaking to remove a drug from the shortage list.

Determining what falls within the statutory definition of ‘biological product’ is an interpretative question that courts, rather than the agency, must resolve, Lilly said in a lawsuit repeatedly citing the US Supreme Court’s June decision in Loper Bright.

Hikma has asked the US Court of Appeals for the Federal Circuit for a full court rehearing of a panel decision that reopened a dispute with Amarin over a skinny-label generic version of Vascepa.

Days after Sandoz received US FDA approval for its Enzeevu aflibercept biosimilar, Regeneron sued alleging that Sandoz failed to follow the “patent dance” set out in the BPCIA framework and claiming infringement of multiple patents on its Eylea.

India prohibits the manufacture and sale of over 150 fixed-dose combinations (FDCs) that lack therapeutic justification and pose potential health risks, though all eyes are on how effectively the regulatory apparatus enforces the ban, not the first in the area, amid legal action. One firm has secured an ad interim ruling for its combination drug.

MSN Laboratories will be unable to move forward with any potential ‘at-risk’ launch for its generic version of Novartis’ Entresto blockbuster in the US for now after the originator won a stay of the firm’s ANDA approval pending appeal from the US Court of Appeals for the District of Columbia Circuit.

Novartis continues to fight tooth and nail to protect its $3bn Entresto brand in the US following the latest FDA approval of a generic version.

But the overall impact of the Supreme Court decision may not seem as “immediate” in 340B compared to other federal programs.

Experts expect litigation challenging Medicare’s definitions of single-source drug and bona fide marketing to get a boost from recent Supreme Court rulings, but overall, the IRA is likely better positioned in a post-Chevron world than older Medicare statutes.

The US Federal Trade Commission’s focus on potential anti-competitive behavior by pharmacy benefit managers picks up steam as Chair Lina Khan approaches the end of her term.

Reading Hikma’s press releases and other public documents made it “at least plausible” that a physician would look to prescribe Hikma’s generic Vascepa product for any of its indications, including the highly-valued, patent-protected cardiovascular indication, the US Federal Circuit decided, reopening a lawsuit against the generics firm.

The unanimous decision that the Alliance for Hippocratic Medicine lacks standing to challenge the FDA’s relaxation of the abortion pill REMS leaves unclear whether the court would have deferred to the agency’s expertise on the merits.

Rahul Rao, the deputy director of the FTC Bureau of Competition, discussed the agency’s thinking on recent pharma deals that it viewed as anti-competitive.

The US Court of Appeals for the Second Circuit afirmed a US district court decision to dismiss purchaser and payer claims linked to so-called “pay-for-delay” agreements with generics firms over nebivolol rivals to Bystolic.