Drug Review Profiles

Vadadustat's review overlapped with two other oral HIF-PH class drugs. Safety issues with FibroGen/AstraZeneca’s roxadustat drew attention during vadadustat's first-cycle review, while GSK’s Jesduvroq labeling and postmarketing requirements informed the second-cycle approval.

The Pink Sheet Drug Review Profile explores the US FDA’s approval of vadadustat to treat anemia in chronic kidney disease patients on dialysis. A complete response letter cited the risk of drug-induced liver injury, but postmarketing data from Japan reassured reviewers.

The Pink Sheet’s Drug Review Profile looks at the timeline behind the development and US FDA approval of Akebia’s Vafseo, a treatment for anemia in chronic kidney disease patients on dialysis.

Pink Sheet’s Drug Review Profile explores Amgen’s Wezlana, an interchangeable biosimilar to Janssen’s Stelara. FDA determined late in the review that switching studies generally would not be needed for interchangeable ustekinumab products, prompting Amgen to seek the designation.

Assigning different suffixes to the prefilled syringe and intravenous formulations of Amgen’s ustekinumab biosimilar could create confusion and would not further the goals of the naming convention, the FDA said in explaining why it departed from its January 2017 final guidance.

Pink Sheet’s Drug Review Profile looks at the timeline behind the development and FDA review of Amgen’s Wezlana (ustekinumab-auub), an interchangeable biosimilar to Janssen’s Stelara.

Pink Sheet’s Drug Review Profile looks at the timeline behind the development and FDA review of CellTrans’ treatment for type 1 diabetes.
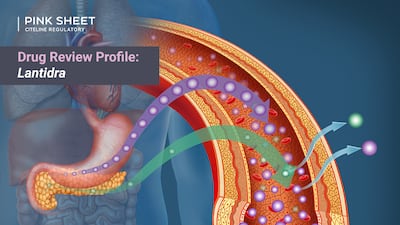
Although the CRL delayed the cell therapy’s approval by almost two years, additional CMC-related testing requested by the FDA ultimately came to be seen as a good thing by sponsor CellTrans because it provided assurance of product consistency.

FDA's review of documents and source data files to assess product efficacy and subject safety was complicated by missing and inconsistent data, the clinical reviewer said, concluding there was insufficient data monitoring during clinical studies or in preparing the documents for regulatory submission.

Despite a host of challenges with donislecel’s development and BLA, the US FDA approved based on clinical data from only 30 patients, using a clinically meaningful insulin independence endpoint and in a more carefully tailored population. Pink Sheet’s Drug Review Profile takes a deep dive into the FDA review of the first approved allogenic pancreas islet cell product.
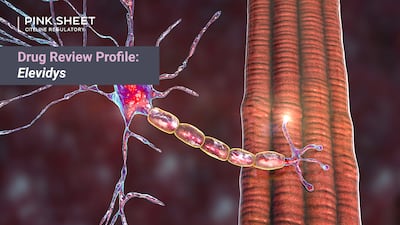
CBER Director Peter Marks urged the review teams for Sarepta’s gene therapy to consider pre- and post-treatment videos showing improved physical function, but clinical and statistical reviewers cited limitations in the patient videos and said they failed to inform whether micro-dystrophin is a suitable surrogate endpoint for accelerated approval.
Timeline of the gene therapy’s clinical development and regulatory review shows agency staff repeatedly questioned the ability of micro-dystrophin to predict clinical benefit in Duchenne muscular dystrophy patients.
When placebo data from the only randomized trial of the Duchenne muscular dystrophy gene therapy were compared with Sarepta’s proffered external control data, the placebo subjects numerically outperformed the external controls, ‘calling into question the comparability of the external control group,’ an FDA statistical reviewer said.

FDA team held several internal meetings on the gene therapy after the 12 May advisory committee, culminating in a teleconference with Sarepta in which the CBER director outlined his accelerated approval decision and reiterated recommendations to modify the ongoing EMBARK trial to better ensure confirmation of benefit. Pink Sheet's Drug Review Profile dives into the story behind the Elevidys review.

The Pink Sheet charts the 13-year journey from Gamida Cell's initial meeting with the US FDA to Omisirge's eventual approval ahead of an extended user fee date.

Pink Sheet’s Drug Review Profile follows the FDA’s navigation of the first expanded umbilical cord blood graft with the first indication grounded in neutrophil recovery and infection incidence data.

Timeline of lecanemab’s clinical development shows how, almost three years after telling Eisai and Biogen that their Phase II results did not support regular or accelerated approval, the FDA review division reversed course following its decision to approve aducanumab on a surrogate endpoint.

With the second amyloid-targeting antibody for treatment of Alzheimer’s, US FDA opted for a class-wide approach to management of amyloid-related imaging abnormalities, but with a product-specific MRI monitoring schedule that differs from that of Aduhelm.

Pink Sheet's Drug Review Profile digs into the FDA memos on Eisai/Biogen’s lecanemab; Phase II clinical efficacy results were reviewed for whether they supported the likelihood of amyloid plaque reduction to predict clinical benefit, rather than whether they directly provided substantial evidence of effectiveness.

Pink Sheet Drug Review Profile looks at how the agency determined that Amylyx’s ‘conceptual basis’ for use of the drug’s two active ingredients to treat ALS was enough to satisfy the combination drug rule; the sponsor also was allowed to submit certain animal studies and drug-drug interaction data during the course of the NDA review.